Product Overview
Hydrochloric Acid, Reagent, ACS
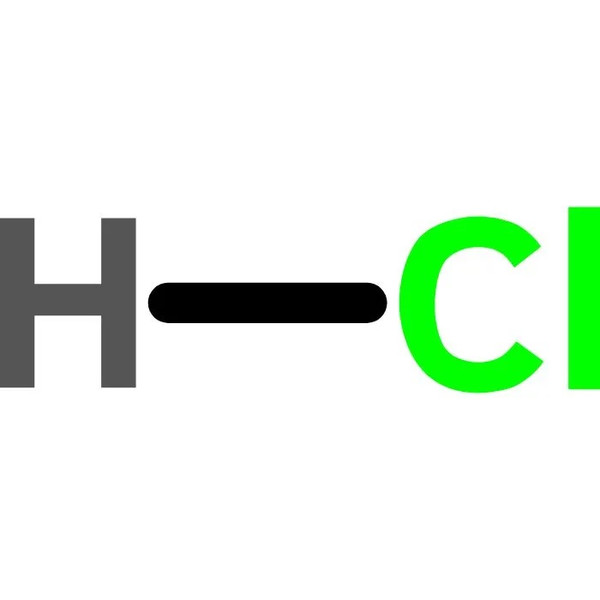
Molecular Formula: HCl
CAS Number: 7647-01-0
Molecular Weight: 34.46
Hydrochloric Acid, Reagent, ACS, is a colorless and clear, highly corrosive, highly pungent solution of hydrogen chloride in water. As an monoprotic acid and one of the six common strong mineral acids, it is the least likely to be subject to an oxidation-reduction reaction. The fact that it is available as a pure reagent make it an excellent acidifying reagent. Hydrochloric acid is also frequently used during chemical analysis to prepare samples for analysis. It reacts with basic compounds, turning them into dissolved chlorides that can then undergo analysis. As an ACS grade Reagent, ScienceLab Hydrochloric Acid is used as the quality standard against which other substances are graded and has met the toughest regulatory standards for quality and pureness.
Synonyms: Muriatic Acid ; Chlorhydric Acid

 WARNING: Cancer and Reproductive Harm - www.P65Warnings.ca.gov
WARNING: Cancer and Reproductive Harm - www.P65Warnings.ca.gov